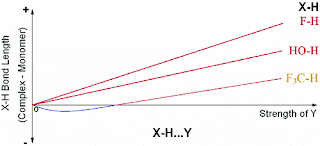
The Wilson's on quality in quantum chemistry
Ken Wilson is very well known for developing the renormalisation group and applying it to critical phenomena and the Kondo problem. There is a long and interesting (but somewhat meandering) interview with him about his career. Amongst various choice tid-bits there is the exchange below about his father, E. Bright Wilson , a pioneer in quantum chemistry. PoS Did your father use computers? Did you know of his models for getting infrared spectra? Did things like that play a role in your thinking? KGW What I remember from discussions with my father was that he used get very wrought up about computational quantum chemists. Garbage in, garbage out. That set me up to spend some time at Ohio State studying quantum chemistry. I had done a little bit towards the end of my stay at Cornell, but took it more seriously while I was at Ohio State. And so then I had to find out from my father who were the good people. And he knew them, he had a list of them. PoS And who did he say were the g