Will burning lots of coal lift people out of poverty?
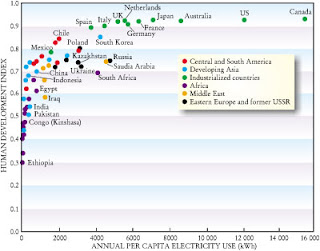
A few months ago I attended a symposium at UQ on Energy in India. The talks can be viewed on Youtube . The one by Alexie Seller is particularly inspiring. In the presentation of Chris Greig he showed a slide similar to that below with the title "Electricity affects Human well being". He did not say it, but sometimes graphs like this are used to make claims such as "the more electricity people consume the better off they will be..." or "the only way to lift people out of poverty is to burn more coal..." Sometimes people show graphs that correlate GDP with energy consumption. But this one is better because it uses the Human Development Index , a multi-dimensional measure of human well being (as it includes life expectancy and education). Two things are very striking about the graph. First, the initial slope is very large. Second, the graph levels off quickly. A little bit of electricity makes a huge difference. If you don't have el